Catalyst: Definition, Types, Mechanism, and Importance in Science
Introduction
In chemistry and biology, the word catalyst plays a central role in understanding how reactions occur. A catalyst is a substance that speeds up the rate of a chemical reaction without being consumed in the process. In other words, a catalyst provides an alternative pathway for a reaction, lowering the activation energy needed for the reaction to occur. This makes reactions faster and more efficient, which is why catalysts are crucial in both industrial processes and biological systems.
This article explores the definition of catalyst, types of catalysts, their mechanisms, industrial applications, biological roles, and significance in modern science.
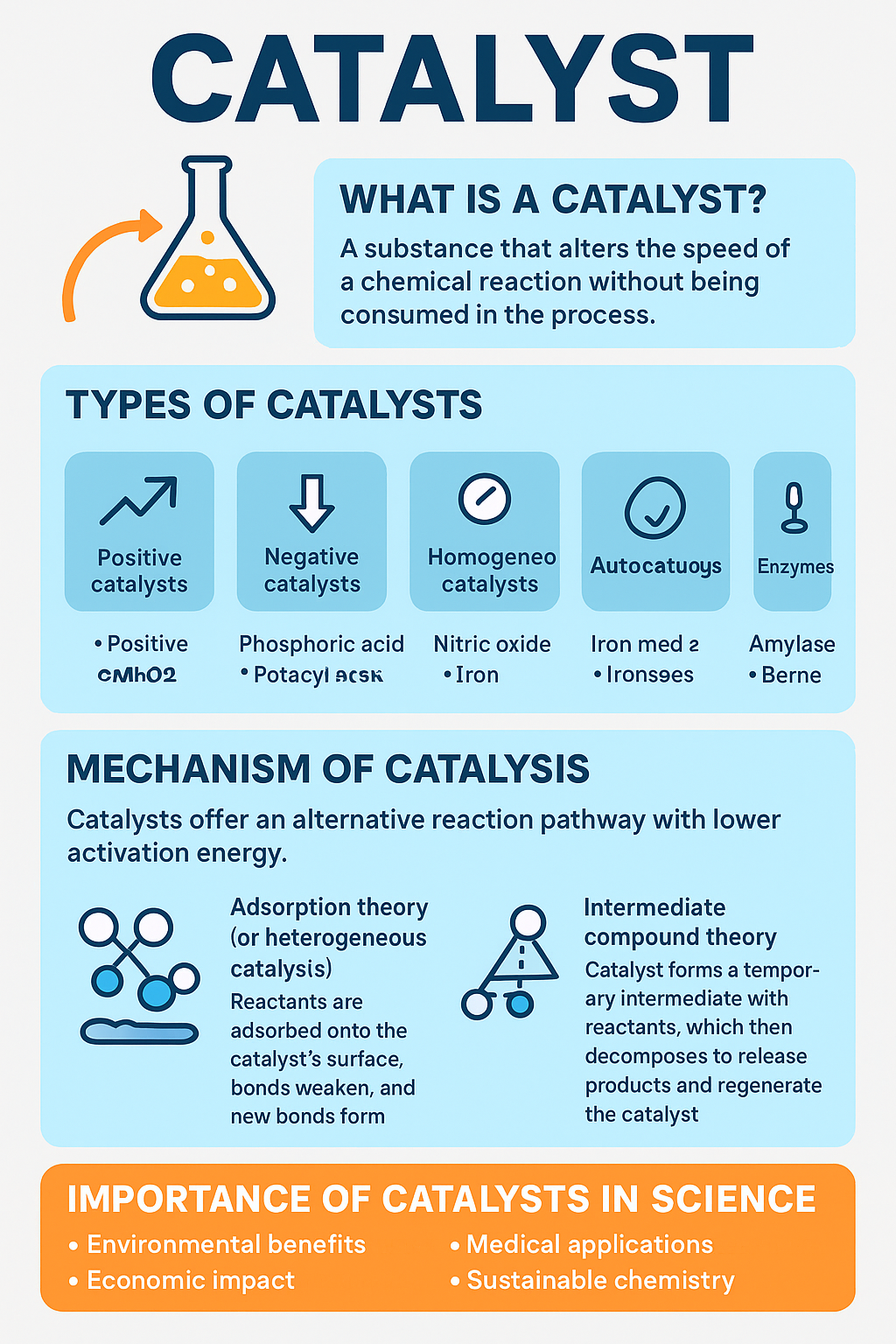
What is a Catalyst?
A catalyst is defined as a substance that alters the speed of a chemical reaction while remaining unchanged chemically and quantitatively at the end of the reaction.
Catalysts lower the activation energy of a reaction.
They do not alter the equilibrium position, but help the system reach equilibrium faster.
A very small amount of catalyst is usually sufficient to trigger significant changes in reaction rates.
Types of Catalysts
Catalysts are broadly classified into different types based on their nature and mode of action:
- Positive Catalysts
These increase the rate of a reaction.
Example: Manganese dioxide (MnO₂) in the decomposition of hydrogen peroxide.
- Negative Catalysts (Inhibitors)
These slow down the rate of a reaction.
Example: Phosphoric acid in the storage of hydrogen peroxide prevents its rapid decomposition.
- Homogeneous Catalysts
The catalyst and reactants exist in the same phase (gas or liquid).
Example: Nitric oxide (NO) in the oxidation of sulfur dioxide during the lead chamber process.
- Heterogeneous Catalysts
The catalyst and reactants exist in different phases.
Example: Iron used in the Haber process (ammonia synthesis).
- Autocatalysts
The reaction product itself acts as a catalyst.
Example: Manganese(II) ions formed during the oxidation of oxalic acid by potassium permanganate.
- Enzymes (Biocatalysts)
Specialized protein catalysts that regulate biological reactions.
Example: Amylase, which breaks down starch into glucose.
Mechanism of Catalysis
Catalysts work by offering an alternative reaction pathway with lower activation energy.
- Adsorption Theory (for heterogeneous catalysis): Reactants are adsorbed onto the catalyst’s surface, bonds weaken, and new bonds form.
- Intermediate Compound Theory (for homogeneous catalysis): Catalyst forms a temporary intermediate with reactants, which then decomposes to release products and regenerate the catalyst.
Industrial Applications of Catalysts
Catalysts are indispensable in modern industries, particularly in chemical and petroleum sectors:
Haber Process: Iron catalyst for ammonia production.
Contact Process: Vanadium(V) oxide for sulfuric acid manufacture.
Hydrogenation of Oils: Nickel catalyst for margarine production.
Catalytic Converters: Platinum, palladium, and rhodium reduce toxic gases in automobiles.
Polymerization: Ziegler-Natta catalysts used for plastic manufacturing.
Biological Importance of Catalysts
In living organisms, catalysts are mainly enzymes:
Digestive enzymes break down complex food (e.g., lipase, protease, amylase).
PhotosynthesisMetabolic enzymes regulate cellular respiration and photosynthesis.
DNA polymerase helps in DNA replication.
Without enzymes, life-sustaining reactions in the body would proceed too slowly to support life.
Characteristics of Catalysts
- Remain unchanged at the end of the reaction.
- Effective in small amounts.
- Do not affect reaction equilibrium, only rate.
- Can be specific – one catalyst works for a specific reaction.
- May be poisoned or deactivated by impurities.
Importance of Catalysts in Science and Society
Environmental Benefits: Catalytic converters reduce car emissions.
Economic Impact: Catalysts reduce energy requirements, saving costs.
Medical Applications: Enzymes are used in drug development and treatments.
Sustainable Chemistry: Catalysis makes chemical processes greener and more efficient.
Discover what a catalyst is, its definition, types, mechanism, and importance in chemistry, biology, and industry. Learn how catalysts speed up reactions without being consumed.
Frequently Asked Questions (FAQ)
- What is a catalyst in simple terms?
A catalyst is a substance that speeds up a reaction without being used up in the process. - How does a catalyst work?
It provides an alternative pathway for the reaction with lower activation energy. - What are the main types of catalysts?
Homogeneous, heterogeneous, autocatalysts, positive, negative, and enzyme catalysts. - Does a catalyst change the equilibrium of a reaction?
No, it only helps the reaction reach equilibrium faster. - Why are catalysts important in industries?
They save energy, time, and resources by speeding up reactions in manufacturing processes. - Can catalysts be reused?
Yes, because they remain unchanged after reactions. - What is an enzyme?
An enzyme is a biological catalyst, usually made of protein, that regulates biochemical reactions. - What is catalytic poisoning?
It occurs when impurities deactivate a catalyst’s efficiency. - What is the difference between homogeneous and heterogeneous catalysis?
Homogeneous catalysts are in the same phase as reactants, while heterogeneous catalysts are in a different phase. - What is an autocatalyst?
A catalyst that is formed as a product of the reaction itself. - Can a catalyst slow down a reaction?
Yes, negative catalysts (inhibitors) slow down reaction rates. - How are catalysts used in environmental protection?
Catalytic converters reduce harmful emissions from vehicles. - Why do enzymes work best at specific temperatures?
Extreme temperatures can denature enzymes and reduce their activity. - What industries use catalysts the most?
Petrochemical, pharmaceutical, polymer, and food processing industries. - Are catalysts always solid?
No, they can be solid, liquid, or gaseous depending on the reaction.
Originally posted 2025-08-19 20:12:17.